Overview
Making a lemon battery is a fun way for students to learn about how batteries work by using common household items like lemons, nails, and pennies.
At a Glance
|
|
Planning Ahead
|
Resources
Lemon or other citrus fruit
|
Notes
|
Instructional Plan
|
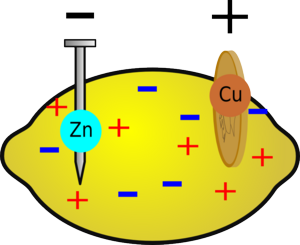
The Science Behind the Experiment
This is an accurate explanation of why this experiment works.
This explanation is easier to understand.
Animation of how a lead battery works. Similar concept, but different chemical reaction.
|
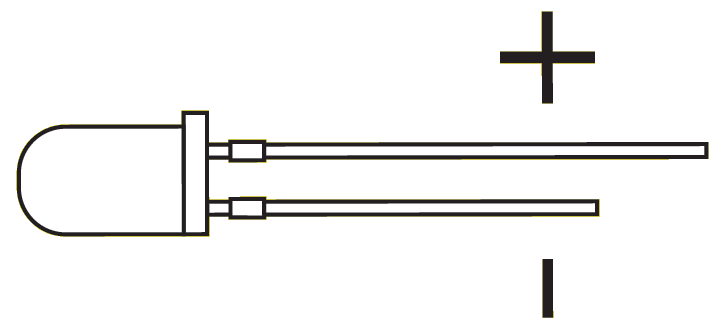
Battery Connections
LED positive and negative leads.
Connect the "copper electrode" to the "long lead" of the LED and the "zinc electrode" to the "short lead" of the LED. Note: If LED doesn't light, flip the leads.
|
Connecting Batteries
If you connect your lemon batteries in series (copper to zinc), you increase the voltage. If your LED does not light at all, try adding batteries in series. Connecting these batteries together in series is like using multiple batteries in a toy. You connect the positive electrode (copper) of one battery to the negative electrode (zinc) of the next battery.
If you connect batteries in parallel, you increase the current. If your LED lights, but is faint, then try increasing the current. You would connect the positive electrode (copper) of one batter to the positive electrode of the next battery. You would connect the negative electrode (zinc) to the negative electrode of the next battery.
|
Step-By-Step Instructions
Theoretically, there are lots of different ways to do this project. You can just google it on the Internet. We were able to successfully run this experiment with the following configuration:
|
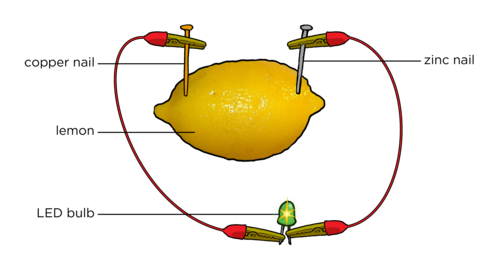
Step 1: Build a Single Battery
|
|
NOTES
We started the experiment using a very large galvanized spike (knowing that would give us a good amount of surface area). Once we got the 3 batteries connected and the LED was lighting, we started replacing the "spike" with smaller galvanized nails. Two nails ended up working. We had hoped to replace the lemon juice with lemons and copper wire with pennies. If we had a volt meter, we could've made the changes based on measurements. We didn't have a volt meter and gave up. |
Optional Penny Nickel Battery Experiment
Here's an optional experiment that works reliably which demonstrates the same concepts. We used pennies and zinc washers. |